Supervised by: Jasmine Lee, BA (Hons) (Cantab.). Jasmine recently graduated from the University of Cambridge having studied Psychological and Behavioural Sciences. She did outreach work for Pembroke College as a student ambassador, and organised TEDx events as the co-president of TEDxCambridgeUniversity. She is currently studying for her masters in Developmental and Educational Psychology at University College London (UCL).
Abstract
This paper summarises findings on genetics, neurobiology, and social factors of anxiety disorders among adolescents, specifically social anxiety disorder (SAD) and generalised anxiety disorder (GAD). Nearly 15 million adult Americans have SAD and 6.8 million have GAD, with onset of symptoms often in adolescence (SingleCare Team, 2022; Gregory et al., 2007). Adolescence is a core risk phase for the development of anxiety symptoms and syndromes, ranging from transient mild symptoms to full-blown anxiety disorders. This article aims to review current literature of anxiety from different fields to create a fuller picture to inform the development of future preventions and treatments.
Introduction
Anxiety has seen a significant increase within the 21st century, and this rings true within adolescents in particular. Hence, it is pertinent that we understand this epidemic, given its increasing prevalence in modern society (Goodwin et al., 2020). Anxiety disorders are defined in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-V) as excessive anxiety and worry (apprehensive expectation), occurring more days than not for at least 6 months, about a number of events or activities (such as work or school performance) (American Psychiatric Association, 2013). Typically, it is difficult for the person with an anxiety disorder to control their worry. Common symptoms include, but are not limited to: restlessness, feeling easily fatigued, difficulties concentrating, and irritability.
Age of Onset and Prevalence
The diagnosis age varies for different types of anxieties. According to research, the age of onset of separation anxiety disorder, specific phobia, and social phobia was on average before 15 years, while agoraphobia, obsessive-compulsive disorder, post traumatic stress disorder, panic disorder, and generalised anxiety disorder had an average onset of between the ages of 21.1 and 34.9 (Lijster et al., 2017). Overall, the average age prevalence for all anxiety disorders was 21.3 years (95% CI 17.46 to 25.07). Anxiety is thus especially prevalent during adolescence. For generalised anxiety disorder (GAD), women are twice as likely to suffer as men (Remes et al., 2016). However, it is important to note that this is the average age of diagnosis; anxiety can happen very late in life due to different factors, but symptoms of anxiety typically begin in adolescence.
Importance of Studying Anxiety
Anxiety is a normal response to challenges. However, with an excessive amount of worries, people’s ability to conduct their obligations would be influenced negatively. By understanding how anxiety develops, we can develop more effective preventions and treatments for adolescents with anxiety.
This paper takes a deep look into anxiety from both biological and psychological viewpoints. The definite causes of anxiety remain unknown: it could potentially be inherited and/or developed later in life. Therefore this paper will discuss possible factors from both genetic and neurobiological perspectives to provide a more comprehensive view. We also discuss the social factors of anxiety, categorised into family, peers, and society. By delving into the concept of anxiety disorders, the authors intend to draw the readers’ attention to this mental illness and realise the importance of research.
Genetics/Epigenetics
Humans are more likely to develop an anxiety disorder if their biological relatives have an anxiety disorder or another mental disorder (McLaughlin et al., 2008). Just as the risk for someone to develop cancer is higher if one of their biological parents has had cancer, it is also similar for many mental disorders; in this review we focus on anxiety disorder. Nevertheless, even if one biological parent has anxiety disorder, the offspring does not necessarily also develop anxiety. Genetics are not the full story: they interact with environmental factors – everything from traumatic events to a parent’s geste – to produce the conditions for anxiety to manifest. There are various ways to study and research the existence of a heritable component to anxiety.
Humans have between 20,000 and 25,000 genes that make up our Deoxyribonucleic acid (DNA). Our DNA determines many of our characteristics, from our eye colour to height. Genes can interact with the environment as well, the study of which is called epigenetics. For example, if someone experiences a traumatic event, their genetic expression may shift to adapt to the trauma, and these epigenetic changes are also sometimes inherited. Although research is still emerging surrounding the genetic influences on mental health, evidence shows that there are genetic predispositions to developing mental illness, including anxiety (Taylor & Hasler, 2018).
Twin Studies
Twin studies have shown that anxiety disorders are moderately heritable. This study is a special type of epidemiological study designed to measure the contribution of genetics as opposed to the environment for a given trait.
In attempting to explain the familial predisposition to panic disorder, most studies have focused on the heritability of physiological characteristics. A less explored possibility is that a psychological predisposition to panic-anxiety sensitivity may also be inherited. Therefore, through the use of a twin group, Stein and colleagues (1999) examined the heritability of anxiety sensitivity. The scores on the Anxiety Sensitivity Index were examined in a group of 176 monozygotic and 158 dizygotic twin groups. Biometrical mode fitting was conducted through use of standard statistical methods. Results demonstrated that the broad heritability estimate of the Anxiety Sensitivity Index as a unifactorial construct was 45% (Stein et al., 1999). Anxiety sensitivity was a product of additive genetic effects and unique environments. This study suggests that the development of panic disorder-anxiety sensitivity may have a heritable component.
Genome-Wide Association Study (GWAS)
Generalised Anxiety Disorder (GAD) is a common anxiety-related diagnosis in adolescents. It is characterised by excessive worry and tension about everyday events that cannot be controlled, and is expressed on most days for at least 6 months to warrant a diagnosis. It should also reach an extent where distress is significant, or where there is difficulty in performing day-to-day tasks. Here we present a genome-wide association study (GWAS) of anxiety sensitivity using a cohort of 230 monozygotic and dizygotic female twins (Davies, 2015). GWASs help scientists identify genes associated with a particular disease or another trait. It is an approach that involves rapidly scanning markers across complete sets of DNA or genomes.
The GWAS showed a significant association for a warrant within the RBFOX1 gene. The full name for the gene is RNA Binding Fox-1 Homolog 1. In this study, it was found out that the RBFOX1 gene may increase the likelihood of developing GAD (Davies, 2015).
Animal Models of Anxiety
Another perspective we could take is using animal models of anxiety in research. The initial observation of Charles Darwin in 1872 that animals and human beings share similar characteristics in the expression of emotion raised the possibility of studying the mechanisms of psychiatric disorders in other mammals. This study can certainly be useful to find out more about the biological bases of anxiety disorders, in order to develop new and efficient pharmacological and behavioural treatments.
A number of anxiety disorder subtypes are considered “stress-related”. A large focus of research has been on the epigenetic and anxiety-like behavioural consequences of stress. The study of epigenetic mechanisms within the context of effects of stressors has provided increasing support for the hypothesis that modifications to gene expression, that are observed as a consequence of these experiences, may involve epigenetic pathways. In particular, variation in DNA methylation and histone modifications has been observed in offspring exposed to prenatal stress, disruption to mother-infant interactions, variation in maternal care, as well as adult social stress. These stressors may account for the deleterious impacts of post-weaning social isolation on the development of rodents (Lukkes et al., 2009).
Animal models of anxiety-related characteristics have provided strong evidence for the role of stress on the epigenetic control of the hypothalamic-pituitary-adrenal (HPA) axis and of stress-responsive brain regions. Chronic variable stress experienced by gestational females has been demonstrated to induce a long-term impact on HPA pathways, including altered gene expression within the hypothalamus. In mice, stress during the 1st week of pregnancy has been found to induce significant impairments in male offspring (Mueller & Bale, 2008). The development of animal models of anxiety and stress has been helpful in increasing our understanding of the pharmacological mechanisms and potential clinical effects of several anxiolytic drugs.
Although anxiety and stress are closely related, they are actually very different as stress occurs as a reaction of one’s body towards the feeling of tension that results from a certain event. When someone feels angry, nervous, irritated or frustrated, it could possibly lead to stress. On the other hand, anxiety is much more complicated than stress, as one may experience overwhelming thoughts and excessive worries for no apparent reason. Some might have constant fear that they will act in a way that will be embarrassing and humiliating. Some may feel restless and anxious, which may make it substantially difficult to get through the day, as one of the many side-effects of anxiety is stress. As such, in studies with animal models, there is no way for us to know whether the animals are anxious or stressed over a given situation. As much as it is relevant and useful to have animal models, given the ethical limitations of human experimentation, further research is required in this field to study the translational relevance of animal models in the context of anxiety disorders.
How Genes and Environmnet Intersect to Create Anxiety
Research suggests there is no single ‘anxiety gene’, but rather numerous genes that interact to cause anxiety, through a biochemical process called DNA methylation (Morris-Rosendahl, 2002). What we eat, how we sleep, the air we breathe, and the stress we face can all alter the expression of our genes. Our genetic code plays a key role, but the environment influences the extent to which those genes will be switched on or off. While your genes are not deterministic, they may account for the difference in coping: one person may respond to a traumatic event with resilience and another may develop an anxiety disorder (Schiele & Domschke, 2017). Potentially, research into the precise mapping of genes may ultimately tell physicians whether a person is prone to developing anxiety just by drawing blood. It is conceivable that someday physicians will be able to distinguish which children are at greatest risk for anxiety disorder and suggest early interventions before the onset of symptoms. However, the ethics of pre-emptive interventions need to be further discussed (Manzini et al., 2021).
Neurobiology
Neuroanatomy of Anxiety
The Limbic System
Anxiety is typically the result of various disruptions on the neuroendocrine, neurotransmitter and neuroanatomical levels (Martin et al., 2009). It is widely believed that anxiety disorders are due to the disruption of the balance of activity in the emotional-processing centres of the brain, better known as the limbic system.
The limbic system is made up of the limbic cortex, comprising the hippocampus, amygdala, and more, which can be seen in Fig 1. The limbic cortex is a region of the limbic system that integrates the sensory, affective and cognitive components of pain. The hippocampus has tonic inhibitory control over the hypothalamic stress-response system, and also plays a role in negative feedback for the hypothalamic stress-response system in the hypothalamic–pituitary–adrenal (HPA) axis. The HPA axis is formed by interactions between the hypothalamus, the pituitary gland and the adrenal glands, and its activation results in a response to stress (Smith & Vale, 2006). Tonic inhibition is where the excitability of all neurons are reduced (Bryson et al., 2020). Negative feedback is a system where mechanisms are fed back into a system to reduce fluctuations caused by disturbances. This is a key process in homeostasis, which helps maintain internal conditions essential for survival (Ashby, 1957). Lastly, the amygdala is responsible for emotions like fear and aggression, allowing us to process emotions and respond to them appropriately. It is located in the medial temporal lobe and comprises the basal amygdala (BA), lateral amygdala (LA), and central nuclei. These three regions have been hypothesised to make up the fear response pathway.
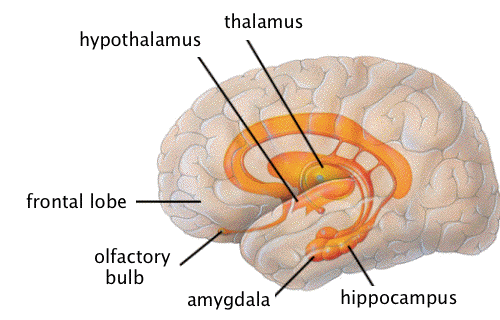
Fig 1: The limbic system
Social Anxiety Disorder
Social anxiety disorder (SAD), sometimes known as social phobia, is a type of anxiety disorder where patients feel an intense, persistent fear of being watched and judged by those around them, specifically in social situations such as speaking in public, and meeting new people (de Lima Osório et al., 2011).
Research into SAD points to the amygdala being activated during the emergence of symptoms. Tasks that involve social cues have been shown to result in hyperreactivity in the amygdala and other limbic areas in patients with SAD. Viewing stimuli that elicit negative emotions, better known as negative affectivity, have also caused patients with SAD to exhibit bilateral amygdala activation. Bilateral amygdala activation also resulted in symptoms of SAD being increasingly severe. Moreover, SAD patients displayed increased subcortical, limbic, and lateral paralimbic activity, suggesting an elevation of their automatic emotional processing. The anterior cingulate cortex (which is associated with decision-making, socially-driven interactions and empathy-related responses), and the prefrontal cortex (associated with executive function) also show decreased activity in SAD patients, suggesting a decrease in their ability for cognitive processing (Lavin et al., 2013).
Neuroendocrine and Neurotransmitter Pathways
The neuroendocrine system is characterised by the interactions between the nervous and endocrine systems – cells involved in the neuroendocrine system release hormones into the bloodstream as a response to stimulation of the nervous system (Frohman, 2009). Neurotransmitters, on the other hand, are chemical messengers that carry chemical signals from one neuron to target cells.
Amino Acid Neurotransmitters
In patients with anxiety disorders, regions of the brain that process emotions exhibit increased activity, which could be the result of a decrease in inhibitory signalling by γ-amino-butyric-acid (GABA). GABA is an inhibitory neurotransmitter, i.e. a chemical messenger, which counteracts the excitatory neurotransmitter glutamate (Lydiard, 2003); it essentially inhibits the effects and symptoms of anxiety in patients. This is done by blocking specific signals in the central nervous system, and is a form of tonic inhibition. There is also an increase in the level of glutamate activity – glutamate is the main excitatory neurotransmitter, working in opposition with GABA and allowing chemical messages to be carried between nerve cells. In comparison with neurotypical subjects, patients with SAD have a 13.2% higher glutamate to creatine ratio in the anterior cingulate cortex. Creatine is a chemical which is used to instantaneously provide energy to tissues with a heightened demand for energy (Avgerinos, 2018). This indicates a positive correlation between the ratio of glutamate to creatine and the severity of SAD symptoms (Martin et al., 2009).
Neuropeptides
Neuropeptides are known to have an effect on social behaviour. Neuropeptides are small proteins, produced by neurons that act on G protein-coupled receptors (GPCR), which are a type of membrane receptor (Merighi, 2017). They are responsible for slow-onset, long-lasting modulation of synaptic transmission. Oxytocin and vasopressin are neuropeptides which are often studied particularly in relation to SAD. Oxytocin, commonly known as the love hormone, has the primary function of facilitating childbirth, and is typically associated with familial and romantic relationships (Magon & Kalra, 2011). Vasopressin regulates blood pressure, blood osmolarity and blood volume (Garrahy & Thompson, 2019). Research by Huber and colleagues (2005) suggested that oxytocin reduces amygdalar activation and inhibits communication between the amygdala and brainstem sites that mediate fear responses. These results from animal studies thus drive the hypothesis that oxytocin can dampen anxiety in humans. Additionally, amygdalar activity has been shown to be influenced by some genetic variants of the central vasopressin and oxytocin receptors, which support the hypothesis of amygdala hyperactivity in SAD.
Corticotropin-Releasing Factors and the Hypothalamic-Pituitary-Adrenal Axis
Corticotropin-releasing factors (CRF) are a type of hormone released by the hypothalamus, and is the main regulator of the HPA axis. Patients with SAD’s HPA axis have been hypothesised to be more sensitive to stimuli, with psychosocial stress resulting in a larger spike in plasma cortisol in comparison to control patients with the same level of cortisol at neutral states. Cortisol is a hormone that regulates the body’s stress response. SAD patients displayed higher cortisol levels in the Trier Social Stress Test (TSST). The TSST is a test used to induce stress, where participants undergo an interview-style presentation, then sit a surprise mental arithmetic test in front of an unresponsive interview panel that does not give feedback or encouragement to the participant. The level of cortisol elevation correlated with increased social avoidance during the approach-avoidance task (Allen et al., 2017).
Functional Neuroanatomy
The amygdala is overactive at rest in primary mood when it encounters the feeling of sadness, but not overactive at rest when the feeling becomes anxiety. People with anxiety, at normal or pathological levels, have an overactive amygdala during symptom provocation instead. Interestingly, the left side of the amygdala is found to be related to sadness while the right side tends to be related to problems of anxiety (Blackmon et al., 2011).
Generalised Anxiety Disorder
As stated above, generalised anxiety disorder (GAD) is a long-term disorder, characterised by excessive, exaggerated anxiety about various everyday activities (Munir & Takov, 2022). Patients may worry and anticipate disaster, be overly concerned about money, health, family, work, etc. This worry is typically difficult to keep under control, and patients tend to expect the worst for no solicited reason.
Brain Anatomy and Neuroimaging
Neuroimaging studies involve observation of the structure and function of the nervous system using imaging technology such as functional magnetic resonance imaging (fMRI), which is used to measure brain activity through changes in blood flow within the brain (Zhang et al., 2020). This technology works off the basis that blood flow increases in regions of the brain that are active (Logothetis et al., 2003).
Structural imaging studies show that patients with GAD typically exhibit high ratios of grey matter to white matter in the upper temporal lobe. Grey matter is a part of the brain where neuronal cell bodies, neuropil (dendrites and unmyelinated axons), glial cells (astrocytes and oligodendrocytes), synapses and capillaries are abundant (Purves et al., 2008). Its function is to process information in the brain. On the other hand, white matter is tissue found between grey matter, which passes messages between areas of grey matter. It mainly contains myelinated axons (Blumenfeld, 2010); myelin is a fatty sheath which facilitates transmission of electrical impulses, and also gives white matter its outwardly whitish colour (Morell & Quarles, 1999). Patients with GAD also have increased amygdalar volume, which may correspond to stress-induced amygdalar hypertrophy, i.e. enlarged amygdalae.
In adolescents with GAD, patients exhibit elevated levels of activity in the ventrolateral prefrontal cortex region (VLPFC), in comparison with neurotypical control patients. VLPFC is a region of the brain that is involved in emotion, reward, motivation, threat detection and fear (Koenigs & Grafman, 2009) (Fig 2). However, VLPFC activity has a negative correlation to the severity of GAD symptoms; hence, it is believed that the increase in VLPFC activity is due to the brain compensating for GAD, rather than it being a cause of GAD. Patients with GAD can also be further split into 2 groups: the first being those with early-life stress, and the second being those without early-life stress. The former group is characterised by a decrease in neuronal viability in the right dorsolateral prefrontal cortex (DLPFC), indicated in Fig 3, whilst the latter group by an increase in neuronal viability within the same region (Martin et al., 2009).
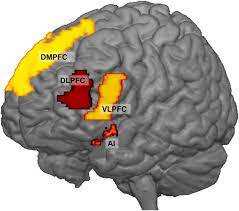
Fig 2: Diagram showing ventrolateral prefrontal cortex (VLPFC)
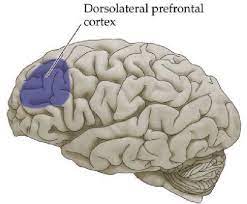
Fig 3: Diagram showing the dorsolateral prefrontal cortex (DLPFC)
fMRI can also be used to analyse the brain activity of patients with GAD. Patients who are in resting conditions tend to have more variable, inconsistent brain activity than neurotypicals, but activities that are anxiety-inducing produce more robust and interpretable fMRI results. In these scenarios, GAD patients were seen to have elevated amygdalar and insular activation. The insula is a small section of grey matter tissue that separates the temporal lobe from the inferior parietal cortex, and is involved in various functions, from sensory and affective processing to high-level cognition (Uddin et al., 2017). Results also showed that severity of GAD symptoms positively correlated with an increase in right amygdala response, which also negatively correlated with activity in the right VLPFC. It is hypothesised that the reason that the amygdala plays such a big role in anxiety disorders is due to it being interconnected with many brain regions that interpret social behaviour (Martin et al., 2009).
Neurotransmitter and Neuroendocrine Signalling: Amino Acid Neurotransmitters
Patients with GAD may exhibit decreased inhibitory neurotransmission, increased excitatory neurotransmission, or a mixture of the both. This may be another reason that the limbic system is hyperactive in GAD patients. In several anxiety disorders, dysregulation of GABA inhibitory neurotransmission is seen. Dysregulation refers to unstable or erratic outputs from neurotransmitters due to the disruption of one or more homeostatic regulatory mechanisms in neurotransmitters (Siever & Davis, 1985). GABA(a) receptor, a receptor that is a ligand-gated ion channel – meaning they open and close when a chemical messenger, like a ligand, binds to it – is seen to be downregulated in patients with GAD. Downregulation is when the amount of GABA(a) receptors is reduced.
Corticotropin-Releasing Factor and the Hypothalamic-Pituitary-Adrenal Axis
Surprisingly, there has been no evidence that indicates increased cortisol or CRF levels in patients with GAD. It may be because CRF and the HPA axis do not play a large role in GAD. However, there have been very few studies that examine HPA axis activity in relation to GAD. Moreover, current literature often examines anxiety disorders in general, yet there is a lack of research into the neurobiology of the developmental course of anxiety in adolescence in particular. Anxiety-related neural networks may be specific to the adolescent brain, hence warranting more research in this area (Xie et al., 2021).
Social Factors of Anxiety Disorders
When trying to understand the reason why different people have different chances of having anxiety disorder, social factors are as important as neurobiological and genetic factors. Social factors are crucial as they shape our social life, which has a significant impact on mental health. This can be explained by Bandura’s Social Learning Theory (1977). This theory states that people can learn through others’ experiences and by observing, unlike in behaviourism which states that you have to personally experience something to learn. This theory relates to anxiety as people do not have to personally experience a situation to have an anxiety for that situation. For anxiety disorders, understanding this first may help to understand the reasons behind it.
Family Influences
Parent-children relationships and the way a child is raised by their parents have been directly associated with anxiety, especially if the parents pose a source of stress and pressure to the child’s life. If the child is raised in an overprotective and controlling environment, this may result in them not being able to develop the necessary skills to cope with stress due to the lack of life experience (Frosch et al., 2019). When a stressful event happens in the child’s life, expectedly, they may become over anxious. In addition, an anxiety-promoting parenting style leads to an insecure attachment style for the child, which may lead to greater anxiety. An insecure attachment style means that the child is unable to use the caregiver as a secure base and feel safe to explore the world (Kerns & Brumariu, 2014). According to Bandura’s social learning theory, a child learns and practises reactions by observing parent reactions. In a stressful situation, a child will copy the behaviour of the parents, such that if the parents react in an over-anxious way, the child will also react over-anxiously. An insecure attachment is thus correlated with increased risk for anxiety disorders (Brumariu & Kerns, 2010). Parents diagnosed with an anxiety disorder may also reflect on the child, once again due to the impact of parental behaviours. A stressed parent may pass their anxiety to the child, creating a threatening environment for the child and causing anxiety. Chapman and colleagues (2022) found that children of parents with anxiety are at greater risk of developing anxiety disorders themselves, yet this is not necessarily so as various treatments are effective in preventing this intergenerational transmission. Moreover, harsh rejection and lack of warmth from parents have been associated with the development of anxiety disorders in children (Brook, 2008). This rejection may cause self-blame and a fear of rejection, further reinforcing an insecure style of attachment. In contrast, a secure parent-child relationship would allow the child to express themselves openly in a stressful situation (e.g. peer pressure) and obtain help to cope with the stressor. Parent-child relationships thus significantly shape children’s emotional development.
In addition to the parent-child relationship, sibling relationships also have a great impact on the development of anxiety. Being bullied by the older sibling, or bullying the younger sibling creates a stressful and discordant environment when growing up (Brook, 2008). Exposure to violence is also highly related to anxiety (Ward et al., 2001). If a child has a sibling with anxiety, they are also likely to observe their anxious behaviours, and imitate them in similar situations. Siblings of children with psychiatric difficulties are thus at greater risk of psychopathology themselves (Ma et al., 2015).
Peer Influences
Peer pressure and social anxiety are directly related to each other. Adolescence is a very vulnerable stage for developing anxiety due to complex changes to the social landscape. Adolescents often make decisions based on others, which is caused by drastic hormonal changes in their bodies and increased social motivation (Ciranka & van den Bos, 2019). In this place of malleability and vulnerability, peers play a significant role in shaping adolescents’ mental health. Bullying is one strong example. A teenager who has been bullied by their friends would most likely lack self confidence, and lack of self confidence may lead to anxiety (O’Moore & Kirkham, 2001). Additionally, negative interactions between the bully and the victim is also a determinant to social anxiety (Pontillo et al., 2019). The defining criteria of SAD is a fear of social situations in which embarrassment and humiliation are possible (Rose & Tadi, 2022). Bullying directly leads to these negative emotions, which is why bullying relates to the development of SAD.
Adolescents are very vulnerable to trauma. A single traumatic event in this period, such as experiences of sexual and physical abuse, may cause the development of anxiety. These adverse events play a significant part in causing social distress for certain environments and potentially the onset of SAD (Bjornsson et al., 2020). People with anxiety disorders were more likely to have experienced childhood physical abuse, which was more common for women than men. However, trauma from sexual abuse is more closely related to panic disorder than anxiety disorders (Stein et al., 1996).
Cultural/Social Influences
The social situation and the cultural environment play a big role in the development of anxiety disorder. Our culture directly shapes our personalities and vulnerabilities. This is because we grow up in our own cultures. Every culture is different in their own ways. It is safe to say that even the definition of anxiety differs between cultures, and this difference also creates a difference in the reported prevalence of anxiety. Cultures are often divided into two types, collectivistic and individualistic cultures. Collectivistic cultures prioritise harmony in a society and value solidarity more, while individualistic cultures value individual’s emotions and goals more (Fatehi et al., 2020). Due to this, more rules and guides to social behaviours are taught in collectivistic cultures to protect the harmony within society. Research has suggested that people from collectivist societies are more accepting of socially reticent and withdrawn behaviours than people from individualistic cultures (Heinrichs et al., 2005). Moreover, participants from collectivistic countries had a higher rate of fear of blushing and social anxiety disorder than individualistic countries. Researchers not only compared collectivistic and individualistic cultures but also searched for a relationship between social anxiety disorder and ethnicity. It has been revealed that black and colored communities had a higher prevalence of anxiety than others (Muris et al., 2005; Vendlinski et al., 2006). This is due to ethnic differences playing a big part in moderating the connection between poor family functioning (marital conflict and lack of warmth) and anxiety (Vendlinski et al., 2006).
Socioeconomic circumstances also play a role in the development of anxiety. This applies more so for adults than adolescents. Schneier and colleagues (1992, cited in Vorcaro et al., 2004) first suggested that anxiety and depression were related to socioeconomic status (SES), whereas other mental disorders were not related. The burden of extreme poverty seems to affect anxiety levels and worrying for the future. This also explains why the countries with higher welfare levels have lower rates of social anxiety disorder. In nations with extreme deprivation, anxiety is especially common. A study in Brazil by Vorcaro et al. (2004) used community samples and collected information through interviews, and data revealed that there was a higher prevalence of anxiety in these samples than in developed countries. SES is thus a significant factor in the development of SAD (Brook & Schmidt, 2008).
Gender Influences
According to the research conducted by the National Institute of Mental Health (NIMH) (2001-2003), GAD affects about 2.7% of American adults, with females experiencing this disorder at a higher rate of 3.4% compared to males at 1.9%. In addition, 2.2% of American teenagers aged 13 to 18 experience general anxiety disorder, in which the percentage was 3.0% for girls, which is double the rate of boys (1.5%). It has also been shown that females diagnosed with one anxiety disorder are more likely than males to be diagnosed with an additional anxiety disorder, bulimia nervosa, or major depressive disorder. Wells (1995, 2009) developed a meta cognitive model of general anxiety disorder accounting for pathological worries. He defined two types of worrying, labelled type I and type II in this model. Type I worry concerns the external and non-cognitive internal events, while type II worry is concerned with negative appraisal of one’s own thought processes. These type II worries are consequences of negative meta cognitive beliefs in the worry process and it has been shown that females suffer from type II worry more. Females may be at greater risk of anxiety as they may believe that worrying is useful in preventing unfortunate events and keeping them aware of warning signs (Bahrami & Yousefi, 2011).
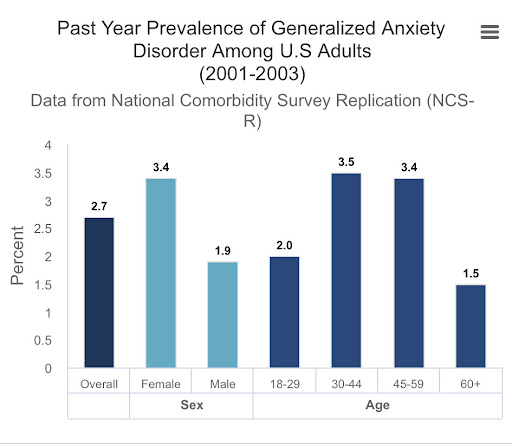
Fig 4. Past Year Prevalence of Generalised Anxiety Disorder Among US Adults (2001~2003)
Conclusion
Summatively, this paper has explored the potential factors for onset of anxiety in adolescence, as well as the importance of study in informing effective preventative measures. We delved into epigenetics, looking at the heritability of anxiety disorders, which is shown through twin studies and animal models. The presence of certain genes have been associated with an increased likelihood of patients having anxiety disorder, although this is not the sole root cause.
Neurobiologically, we looked into how the limbic system contains many regions of the brain that have high levels of association with anxiety prevalence. SAD and GAD were both studied; we looked at the significance of neuroendocrine and neurotransmitter pathways, and how different chemicals play a role in inhibiting or activating regions of the brain that positively correlate with increased symptoms of SAD or GAD. Brain imaging studies are also included, where different levels of activities within certain regions of the brain have correlations with symptom severity.
This article also goes further and explains other significant factors like family, peer, culture, socioeconomic and gender influences. These influences are broad and complex, with family influences not only limited to parent-child relationships but also includes sibling-child relationships, and collectivism and individualism in cultures also having a potential effect. We looked into both ethnic and socioeconomic factors which play a significant albeit small part in the development of SAD.
As an extension to this research paper, it may be interesting to explore the role of genetics in neurobiological causes of anxiety. Given the immense complexity of epigenetics, it may be worthwhile to deepen our understanding in this topic. Additionally, we could perform primary data collection by surveying adolescents about their perception of anxiety and its prevalence within adolescents. Given this study’s value of incorporating perspectives from various fields, future studies could further endeavour to combine the different levels to create a comprehensive model of anxiety.
References
Achterberg, M., van Duijvenvoorde, A., van IJzendoorn, M. H., & Bakermans-kranenburg, M. (2020). ROIs in the left hemisphere. The VLPFC and AI ROIs are bilateral; 3D nifti files of the ROIs are accessible through the OSF [Photograph]. Longitudinal Changes in DLPFC Activation during Childhood Are Related to Decreased Aggression Following Social Rejection. https://www.researchgate.net/figure/ROIs-in-the-left-hemisphere-The-VLPFC-and-AI-ROIs-are-bilateral-3D-nifti-files-of-the_fig2_340323169
Allen, A. P., Kennedy, P. J., Dockray, S., Cryan, J. F., Dinan, T. G., & Clarke, G. (2017). The Trier Social Stress Test: Principles and practice. Neurobiology of Stress, 6, 113–126. https://doi.org/10.1016/j.ynstr.2016.11.001
Al-Thaqib, A., Baradi, R. S., & Khattak, A. H. (2018). Image of the location of the (left) Dorsolateral Prefrontal Cortex in the brain. [Photograph]. An Evaluation of the Comparative Effectiveness of tDCS Treatments for Depression in Saudi Arabia. https://www.researchgate.net/publication/340920818_An_Evaluation_of_the_Comparative_Effectiveness_of_tDCS_Treatments_for_Depression_in_Saudi_Arabia
American Psychiatric Association. (2013). Diagnostic and statistical manual of mental disorders (5th ed.). https://doi.org/10.1176/appi.books.9780890425596
Aron, Robbins, & Poldrack. (2004). Ventrolateral Prefrontal Cortex. Science Direct. https://www.sciencedirect.com/topics/neuroscience/ventrolateral-prefrontal-cortex
Ashby, W. R. (1956). An introduction to cybernetics. John Wiley and Sons. https://doi.org/10.5962/bhl.title.5851
Avgerinos, K. I., Spyrou, N., Bougioukas, K. I., & Kapogiannis, D. (2018). Effects of creatine supplementation on cognitive function of healthy individuals: A systematic review of randomized controlled trials. Experimental Gerontology, 108, 166–173. https://doi.org/10.1016/j.exger.2018.04.013
Bandura, A. (1977). Social learning theory. Englewood Cliffs, N.J: Prentice Hall.
Bahrami, F., & Yousefi, N. (2011). Females are more anxious than males: a metacognitive perspective. Iranian Journal of Psychiatry and Behavioural Sciences, 5(2), 83–90. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3939970/
Bjornsson, A. S., Hardarson, J. P., Valdimarsdottir, A. G., Gudmundsdottir, K., Tryggvadottir, A., Thorarinsdottir, K., Wessman, I., Sigurjonsdottir, Ó., Davidsdottir, S., & Thorisdottir, A. S. (2020). Social trauma and its association with posttraumatic stress disorder and social anxiety disorder. Journal of Anxiety Disorders, 72, 102228. https://doi.org/10.1016/j.janxdis.2020.102228
Blumenfeld, H. (2010). Neuroanatomy through Clinical Cases (2nd ed.). Sinauer Associates is an imprint of Oxford University Press.
Boeree, C. G. (2009). The Limbic System [Photograph]. The Emotional Nervous System. http://webspace.ship.edu/cgboer/limbicsystem.html
Blackmon, K., Barr, W. B., Carlson, C., Devinsky, O., DuBois, J., Pogash, D., Quinn, B. T., Kuzniecky, R., Halgren, E., & Thesen, T. (2011). Structural evidence for involvement of a left amygdala-orbitofrontal network in subclinical anxiety. Psychiatry Research, 194(3), 296–303. https://doi.org/10.1016/j.pscychresns.2011.05.007
Brook, C. A., & Schmidt, L. A. (2008). Social anxiety disorder: a review of environmental risk factors. Neuropsychiatric Disease and Treatment, 4(1), 123–143. https://doi.org/10.2147/ndt.s1799
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2515922/
Brumariu, L. E., & Kerns, K. A. (2010). Parent-child attachment and internalising symptoms in childhood and adolescence: a review of empirical findings and future directions. Development and Psychopathology, 22(1), 177–203. https://doi.org/10.1017/S0954579409990344
Bryson, A., Hatch, R. J., Zandt, B. J., Rossert, C., Berkovic, S. F., Reid, C. A., Grayden, D. B., Hill, S. L., & Petrou, S. (2020). GABA-mediated tonic inhibition differentially modulates gain in functional subtypes of cortical interneurons. Proceedings of the National Academy of Sciences, 117(6), 3192–3202. https://doi.org/10.1073/pnas.1906369117
Campos, A., Fogaca, M., Aguiar, D., & Guimaraes, F. (2013). Animal models of anxiety disorders and stress.https://pubmed.ncbi.nlm.nih.gov/24271222/#:~:text=The%20development%20of%20animal%20models,induced%20by%20approach%2Davoidance%20situations
Chapman, L., Hutson, R., Dunn, A., Brown, M., Savill, E., & Cartwright-Hatton, S. (2022). The impact of treating parental anxiety on children’s mental health: An empty systematic review. Journal of Anxiety Disorders, 88, 102557. https://doi.org/10.1016/j.janxdis.2022.102557
Ciranka, S., & van den Bos, W. (2019). Social influence in adolescent decision-making: A formal framework. Frontiers in Psychology, 10(1915). https://doi.org/10.3389/fpsyg.2019.01915
Davies, M. N., Verdi, S., Burri, A., Trzaskowski, M., Lee, M., Hettema, J. M., Jansen, R., Boomsma, D. I., & Spector, T. D. (2015). Generalised Anxiety Disorder–A Twin Study of Genetic Architecture, Genome-Wide Association and Differential Gene Expression. PloS one, 10(8), e0134865. https://doi.org/10.1371/journal.pone.0134865
de Lima Osório, F., Crippa, J. A. , Hallak, J. E. C. , & Loureiro, S. R. (2011). Social Anxiety Disorder, Fear of Public Speaking, and the use of Assessment Instruments. In (Ed.), Different Views of Anxiety Disorders. IntechOpen. https://doi.org/10.5772/18629
Fatehi, F., Samadbeik, M., & Kazemi, A. (2020). What is Digital Health? Review of Definitions. Studies in Health Technology and Informatics, 275, 67–71. https://doi.org/10.3233/SHTI200696
Frosch, C. A., Schoppe-Sullivan, S. J., & O’Banion, D. D. (2021). Parenting and Child Development: A Relational Health Perspective. American Journal of Lifestyle Medicine, 15(1), 45–59. https://doi.org/10.1177/1559827619849028
Frohman, L. A. (2009). Neuroendocrinology. In L. R. Squire (Ed.), Encyclopedia of Neuroscience (pp. 351–354). Academic Press. https://doi.org/10.1016/B978-008045046-9.01184-0
Garrahy, A., & Thompson, C. J. (2019). Encyclopedia of Endocrine Diseases (Second Edition) ((Second Edition) ed.). Academic Press.
Goodwin, R. D., Weinberger, A. H., Kim, J. H., Wu, M., & Galea, S. (2020). Trends in anxiety among adults in the United States, 2008-2018: Rapid increases among young adults. Journal of Psychiatric Research, 130, 441–446. https://doi.org/10.1016/j.jpsychires.2020.08.014
Gregory, A. M., Caspi, A., Moffitt, T. E., Koenen, K., Eley, T. C., & Poulton, R. (2007). Juvenile mental health histories of adults with anxiety disorders. The American Journal of Psychiatry, 164(2), 301–308. https://doi.org/10.1176/ajp.2007.164.2.301
Heinrichs, N., Rapee, R. M., Alden, L. A., Bögels, S., Hofmann, S. G., Oh, K. J., & Sakano, Y. (2006). Cultural differences in perceived social norms and social anxiety. Behaviour Research and Therapy, 44(8), 1187–1197. https://doi.org/10.1016/j.brat.2005.09.006
Huber, D., Veinante, P., & Stoop, R. (2005). Vasopressin and oxytocin excite distinct neuronal populations in the central amygdala. Science (New York, N.Y.), 308(5719), 245–248. https://doi.org/10.1126/science.1105636
Kerns, K. A., & Brumariu, L. E. (2014). Is insecure parent–child attachment a risk factor for the development of anxiety in childhood or adolescence? Child Development Perspectives, 8(1), 12–17. https://doi.org/10.1111/cdep.12054
Koenigs, M., & Grafman, J. (2009). The functional neuroanatomy of depression: distinct roles for ventromedial and dorsolateral prefrontal cortex. Behavioural Brain Research, 201(2), 239–243. https://doi.org/10.1016/j.bbr.2009.03.004
Lavin, C., Melis, C., Mikulan, E., Gelormini, C., Huepe, D., & Ibañez, A. (2013). The anterior cingulate cortex: an integrative hub for human socially-driven interactions. Frontiers in Neuroscience, 7, 64. https://doi.org/10.3389/fnins.2013.00064
Lijster, J. M., Dierckx, B., Utens, E. M., Verhulst, F. C., Zieldorff, C., Dieleman, G. C., & Legerstee, J. S. (2017). The Age of Onset of Anxiety Disorders. Canadian Journal of Psychiatry. Revue canadienne de psychiatrie, 62(4), 237–246. https://doi.org/10.1177/0706743716640757
Logothetis N. K. (2003). The underpinnings of the BOLD functional magnetic resonance imaging signal. The Journal of Neuroscience : the Official Journal of the Society for Neuroscience, 23(10), 3963–3971. https://doi.org/10.1523/JNEUROSCI.23-10-03963.2003
Lukkes, J. L., Watt, M. J., Lowry, C. A., & Forster, G. L. (2009). Consequences of post-weaning social isolation on anxiety behaviour and related neural circuits in rodents. Frontiers in Behavioural Neuroscience, 3, 18. https://doi.org/10.3389/neuro.08.018.2009
Lydiard R. B. (2003). The role of GABA in anxiety disorders. The Journal of Clinical Psychiatry, 64 Suppl 3, 21–27.
Ma, N., Roberts, R., Winefield, H., & Furber, G. (2015). The prevalence of psychopathology in siblings of children with mental health problems: a 20-year systematic review. Child Psychiatry and Human Development, 46(1), 130–149. https://doi.org/10.1007/s10578-014-0459-1
Magon, N., & Kalra, S. (2011). The orgasmic history of oxytocin: Love, lust, and labour. Indian Journal of Endocrinology and Metabolism, 15(7), 156. https://doi.org/10.4103/2230-8210.84851
Manzini, A., Jones, E., Charman, T., Elsabbagh, M., Johnson, M. H., & Singh, I. (2021). Ethical dimensions of translational developmental neuroscience research in autism. Journal of Child Psychology and Psychiatry, and Allied Disciplines, 62(11), 1363–1373. https://doi.org/10.1111/jcpp.13494
Martin, E. I., Ressler, K. J., Binder, E., & Nemeroff, C. B. (2009). The neurobiology of anxiety disorders: brain imaging, genetics, and psychoneuroendocrinology. The Psychiatric Clinics of North America, 32(3), 549–575. https://doi.org/10.1016/j.psc.2009.05.004
McLaughlin, K. A., Behar, E., & Borkovec, T. D. (2008). Family history of psychological problems in generalised anxiety disorder. Journal of Clinical Psychology, 64(7), 905–918. https://doi.org/10.1002/jclp.20497
Morrell, P. & Quarles, R.H. (1999). Myelin Formation, Structure and Biochemistry. In: Siegel, G.J., Agranoff, B.W., Alber, R.W., Risher, S.K. and Uhler, M.D., Eds., Basic Neurochemistry, 6th Edition, Raven Press, New York, 70-93.
Morris-Rosendahl, D. (2022). Are there anxious genes?https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3181683/
Merighi, A. (2017). Neuropeptides and Coexistence. Reference Module in Neuroscience and Biobehavioral Psychology.
Mueller, B. R., & Bale, T. L. (2008). Sex-specific programming of offspring emotionality after stress early in pregnancy. The Journal of Neuroscience : the Official Journal of the Society for Neuroscience, 28(36), 9055–9065. https://doi.org/10.1523/JNEUROSCI.1424-08.2008
Munir, S., & Takov, V. (2022). Generalized Anxiety Disorder. StatPearls Publishing LLC.
Muris, P. (2006). The pathogenesis of childhood anxiety disorders: Considerations from a developmental psychopathology perspective. International Journal of Behavioral Development, 30(1), 5–11. https://doi.org/10.1177/0165025406059967
O’Moore, M., & Kirkham, C. (2001). Self-esteem and its relationship to bullying behaviour. Aggressive Behaviour, 27(4), 269–283. https://doi.org/10.1002/ab.1010
Pontillo, M., Tata, M. C., Averna, R., Demaria, F., Gargiullo, P., Guerrera, S., Pucciarini, M. L., Santonastaso, O., & Vicari, S. (2019). Peer Victimisation and Onset of Social Anxiety Disorder in Children and Adolescents. Brain Sciences, 9(6), 132. https://doi.org/10.3390/brainsci9060132
Purves, D. (2008). Neuroscience, Fourth Edition (4th ed.). Sinauer Associates, Inc.
Remes, O., Brayne, C., van der Linde, R., & Lafortune, L. (2016). A systematic review of reviews on the prevalence of anxiety disorders in adult populations. Brain and Behaviour, 6(7), e00497. https://doi.org/10.1002/brb3.497
Rose, G. M., & Tadi, P. (2021). Social Anxiety Disorder. In StatPearls. StatPearls Publishing.
Schiele, M. A., & Domschke, K. (2018). Epigenetics at the crossroads between genes, environment and resilience in anxiety disorders. Genes, Brain, and Behaviour, 17(3), e12423. https://doi.org/10.1111/gbb.12423
Siever, L. J., & Davis, K. L. (1985). Overview: toward a dysregulation hypothesis of depression. PubMed. https://pubmed.ncbi.nlm.nih.gov/2862799/
SingleCare Team. (2022). Anxiety statistics in the U.S. 2022. The Checkup. https://www.singlecare.com/blog/news/anxiety-statistics/.
Smith, S. M., & Vale, W. W. (2006). The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues in Clinical Neuroscience, 8(4), 383–395. https://doi.org/10.31887/dcns.2006.8.4/ssmith
Stein, M. B., Walker, J. R., Anderson, G., Hazen, A. L., Ross, C. A., Eldridge, G., & Forde, D. R. (1996). Childhood physical and sexual abuse in patients with anxiety disorders and in a community sample. The American Journal of Psychiatry, 153(2), 275–277. https://doi.org/10.1176/ajp.153.2.275
Stein, M. B., Jang, K. L., & Livesley, W. J. (1999). Heritability of anxiety sensitivity: a twin study. The American Journal of Psychiatry, 156(2), 246–251. https://doi.org/10.1176/ajp.156.2.246
Taylor, B. J., & Hasler, B. P. (2018). Chronotype and Mental Health: Recent Advances. Current Psychiatry Reports, 20(8), 59. https://doi.org/10.1007/s11920-018-0925-8
Uddin, L. Q., Nomi, J. S., Hébert-Seropian, B., Ghaziri, J., & Boucher, O. (2017). Structure and Function of the Human Insula. Journal of Clinical Neurophysiology, 34(4), 300–306. https://doi.org/10.1097/wnp.0000000000000377
Vendlinski, M., Silk, J. S., Shaw, D. S., & Lane, T. J. (2006). Ethnic differences in relations between family process and child internalising problems. Journal of Child Psychology and Psychiatry, and Allied Disciplines, 47(9), 960–969. https://doi.org/10.1111/j.1469-7610.2006.01649.x
Vorcaro, C.M., Rocha, F.L., Uchoa, E., Lima-Costa, M.F. (2004) The burden of social phobia in a Brazilian community and its relationship with socioeconomic circumstances, health status and use of health services: the Bambuí study. Int J Soc Psychiatry. 50(3):216-26. https://pubmed.ncbi.nlm.nih.gov/15511115/
Ward, C. L., Flisher, A. J., Zissis, C., Muller, M., & Lombard, C. (2001). Exposure to violence and its relationship to psychopathology in adolescents. Injury Prevention : Journal of the International Society for Child and Adolescent Injury Prevention, 7(4), 297–301. https://doi.org/10.1136/ip.7.4.297
Wells, A. (1995). Meta-Cognition and Worry: A Cognitive Model of Generalised Anxiety Disorder. Behavioural and Cognitive Psychotherapy, 23(3), 301-320. doi:10.1017/S1352465800015897
Xie, S., Zhang, X., Cheng, W., & Yang, Z. (2021). Adolescent anxiety disorders and the developing brain: comparing neuroimaging findings in adolescents and adults. General Psychiatry, 34(4), e100411. https://doi.org/10.1136/gpsych-2020-100411
Zhang, J., Chen, K., Wang, D., Gao, F., Zheng, Y., & Yang, M. (2020). Editorial: Advances of Neuroimaging and Data Analysis. Frontiers in Neurology, 11. https://doi.org/10.3389/fneur.2020.00257