Supervised by: Yuhui Zhou, BA (Hons). Yuhui is a 5th year medical student at the University of Cambridge. She gained a First class degree in her intercalated year studying Pathology. She has an interest in Cancer & Immunology and has been awarded a Wellcome Trust Biomedical Vacation Scholarship to study host responses to infection.
Introduction
Since COVID-19 hit the world stage in early 2020, more than 503 million people have contracted the virus, with around 6,220,970 fatalities at the time of writing (April 2022) (1). Over the course of the past two years, many different companies have shifted their focus on developing vaccines for the virus, hoping to significantly decrease the fatality rate. There are now a variety of approved vaccines, classified by different technological platforms ranging from traditional live-attenuated vaccines (Oxford-Astrazeneca), which stimulates a specific immune response, to newer mRNA vaccines (Pfizer). The focus of this review will be on the Pfizer and Astrazeneca vaccines, both of which take different approaches to immunising the population. This has resulted in different public reactions towards each during their time in use.
Based on the U.S. Centres for Disease Control and Prevention (CDC), the definition of the term vaccine effectiveness is: “a measure of how well vaccination protects people against outcomes such as infection, symptomatic illness, hospitalisation, and death” (3). The acceptance rate of the vaccines are affected by numerous factors, like knowledge, vaccine efficacy, safety and personal experiences (4). This paper examines the correlation between effectiveness of the vaccine and its acceptance to determine whether vaccine effectiveness is the most influential factor.
The Pfizer vaccine is the most common vaccine that has been rolled out in many countries, since being approved in December 2020 by the UK government (5). As of 20th April 2022, 603.89 million doses have been administered across the world, making it by far the most popular vaccine across the world, most likely because of its high efficacy of 95% against symptomatic COVID-19 infections (6). It is an mRNA-based vaccine, so uses a newer approach to provide immunity against the virus. These vaccines introduce a strand of mRNA that is usually obtained from the virus’s outer membrane. The body’s immune cells use this mRNA blueprint to produce the viral protein, which in turn triggers a humoral response, producing specialised protein structures called antibodies (7). Preliminary laboratory studies show that it is more effective against the newer variants – including Omicron – than Astrazeneca (8).
The Oxford/AstraZeneca (ChAdOx1-S) COVID-19 vaccine was validated by the World Health Organisation (WHO) on 15th February 2021 (following its approval by the British government on 30th December 2020), giving the world assurance that it meets international standards of safety, efficacy and manufacturing (9, 5). The efficacy of Astrazeneca is only 76% against symptomatic COVID-19 infection, 19% lower than Pfizer. Astrazeneca is a live-attenuated vaccine in which a live but weakened virus is directly introduced into the bloodstream. Here, the body’s immune system will identify the virus as foreign and will work to create antibodies for the disease. Therefore, the purpose of this review is to analyse the correlation between the acceptance rate and the efficacy of vaccination, by comparing these two common vaccines (10).
Effectiveness of the vaccines
Neither Pfizer nor Astrazeneca prevent the vaccinated individual from contracting COVID-19, but research has found that it decreases the severity of it. The Pfizer vaccine has a higher efficacy rate at 95%, while Astrazeneca has an efficacy of 88% after two doses, thus making it a more popular choice of vaccine around the world (8). In a study conducted in North-West London between 12th January 2021 and 9th March 2021, published as an article in The Lancet Discovery Science eMedicine Journal, it was found that after having two doses of the Astrazeneca vaccine, the amount of COVID-19 related deaths decreased from 25/1000 to around 2/1000, therefore proving that Astrazeneca is still effective, even if it is not as statistically effective as Pfizer (8).
The same study also concluded that the effectiveness of both vaccines were higher in older age groups – the hazard ratios were found to decrease at advanced age following vaccination. The study showed that Pfizer was more effective in middle-aged individuals, however, and then decreased at a certain age. Likewise, although Pfizer helped reduce the severity of infection, Astrazeneca proved to be a better vaccine for the elderly population of London (8). Of note, however, is that this study was conducted in 2021, where the world was in the early stages of vaccine rollout, putting it at a disadvantage compared to more recent studies where the results would be more up-to-date and reliable.
In another case-control study of 150,000 adults aged 70+ between December 2020 and February 2021, it was concluded that for the elderly age group, Astrazeneca was the better vaccine as it helped to decrease the chance of contracting COVID-19 more than Pfizer, along with the amount of hospitalisations and deaths due to this virus. It found that Pfizer reached an effectiveness of 75% around a month after the first dose in the elderly population, however, Astrazeneca proved to be more effective with an effectiveness of 80% after a single dose, and was 85% effective at preventing COVID-19 related deaths (9).
The side-effects of the vaccines can be compared to see if it has any correlation with the effectiveness of the vaccine. A prospective, observational study was conducted between December 2020 and March 2021, where the “proportion and probability of self-reported systematic and local side-effects within 8 days of vaccinations in individuals using the COVID-19 symptom Study app” were reported (10). The study found that systematic side-effects were more common in people who took the Pfizer vaccine, with 33.7% of them experiencing systematic symptoms of any kind, compared to only 22% of people who took the Astrazeneca vaccine. However, if the individuals took Astrazeneca, they were more likely to get local side effects where those who took Pfizer weren’t. For example, 44.1% of individuals experienced pain and swelling of their arm, while only 24.6% of those who took Pfizer experienced those symptoms. The study also found that local adverse side effects were equally less commonly reported after the second dose of Astrazeneca and Pfizer (10).
There have been severe cases where individuals suddenly develop autoimmune diseases after one of their doses have been given. While most side effects of the vaccine were mild and temporary, a very small fraction of the individuals who received the Pfizer vaccine experienced longer lasting symptoms, which could be because of the mRNA which was taken out of the nucleus and could have left some antigens exposed, which could lead to the immune system attacking itself (4). While Astrazeneca didn’t appear to have long-term side effects, such as autoimmune diseases, the roll-out of the vaccine was temporarily halted in some European countries, as it was reported to be causing blood clots in some individuals.
According to the WHO, herd immunity is ‘the indirect protection from an infectious disease that happens when a population is immune either through vaccination or immunity developed through previous infection’ (11). The concept of herd immunity is the same for all vaccines, be it mRNA or viral vectors; that is, if a large number of a population is vaccinated, then the transmission of any disease will significantly decrease as the rate of transmission would, itself, decrease. The percentage of the population that must be vaccinated varies for each infection; for some diseases like measles, it needs to be as high as 90% of the population. While it is not certain what this percentage is for COVID-19, it is estimated to be slightly lower, at 75-80%. However, this could vary due to new variants (12).
Overall, the difference between the two vaccines depends on the efficacy of the vaccine and how it changes over time. The efficacy of the Pfizer vaccine was found to be 95% after two doses, but it was found that this may wane over time, though not by any meaningful amount. The efficacy of the Astrazeneca vaccine, though, was found to be significantly lower at 76% after two doses. This did increase, however, over a 12-week period to 82% (13, 14).
Acceptance of the vaccines
The efficacy of the various COVID-19 vaccines is clearly an important issue in terms of managing the negative effects of the pandemic and, ultimately, saving lives. However, this paper aims to consider how important the efficacy is in determining vaccine acceptance and uptake.
A systematic search of the peer-reviewed English survey literature indexed in PubMed was completed on 25 December 2020. At that time, some countries had a low COVID-19 vaccine acceptance rate, for example, Russia (54.9%), Poland (56.3%) and the US (56.9%) (15). Vaccine hesitancy is a major challenge in successfully controlling a pandemic. It results in a delayed rollout of vaccines that could put countless millions at risk. There are numerous factors that lead to this hesitancy, which is classified into complacency and confidence. Complacency denotes the low perception of the disease risk. The low mortality rate of about 0.5% to 5%, resulted in complacency. Some people did not see the urgency in taking the vaccine, which they believed might have had a higher risk than the virus at the time. They believed that our natural immune system would be able to overcome the sickness. Confidence refers to the trust in vaccination safety and effectiveness. The data collected from the vaccines are from the recent two years. There is not enough investigation into the vaccines’ long-term effects. Despite guarantees about the safety of the vaccines, many were concerned about the undiscovered side-effects they might have. The durability of the vaccines and their efficacy towards future variants are still somewhat unclear, especially with many countries not offering boosters. With the lack of information and knowledge, people were hesitant to get vaccinated during the initial rollout, hence the low acceptance rate (16).
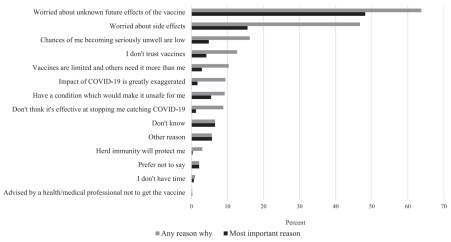
Figure 1: Reasons leading to vaccine hesitancy (n=375)
Although lack of knowledge about the vaccine contributed to the low acceptance rate, it is unclear whether more information about the vaccine would further discourage people from taking it. One example of this is in the phenomenon of mass media misinformation regarding adverse events reported after COVID-19 vaccination. The mass media would put anaphylaxis after COVID-19 vaccinations on their headlines, arousing fear and concerns in the public. However, such cases are very rare, with about 5 in a million cases. It can also be effectively and immediately treated. Reports of adverse events following vaccination, including deaths, leads to further distrust in the vaccine as people believe it might be causing health problems. Multiple studies and reviews of data from vaccine safety monitoring systems continue to show that vaccines are safe and effective. Results from these studies have comforted the public and increased their trust in the vaccines. People who work in healthcare industries, like virologists and doctors, have more knowledge on COVID-19 and its vaccines. A study showed a higher acceptance of vaccination among healthcare workers, at 76.98%, than non-healthcare workers at 56.19%. The rates of vaccine hesitance and resistance are also lower than those of non-healthcare workers. This justifies that more knowledge can affect the acceptance rate of vaccination in both directions (16).
The acceptance rate between Astrazeneca and Pfizer are different across regions and generations. Astrazeneca is a type of viral vector vaccine (17). Such a method has been around for a few decades, allowing us to collect more data and conduct research on such vaccines. Conversely, Pfizer is the first mRNA vaccine, which uses a new approach to treating viruses (17). The long-term effects of such a method might be unclear to many. Notwithstanding the lower effectiveness, some people prefer to play it safe, choosing Astrazeneca instead since they have more trust in its method. Studies show that younger generations are more likely to reject vaccination. A few possible reasons are the lack of knowledge, and urgency. Since the infection and mortality rate in elderly people are higher, due to their weakened immune system, they might see an urgency in taking the vaccine. However, the younger generations might prefer to rely on their own natural immune system to fight COVID-19 (16).
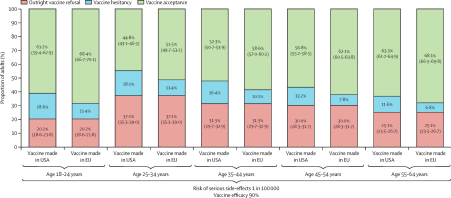
Figure 2: Vaccine hesitancy and acceptance between different age groups
Astrazeneca seems to be more widely accepted around the world, despite its lower effectiveness of just 76% compared to Pfizer’s 95%. Most countries have chosen Astrazeneca, compared to other vaccines. This might be because Astrazeneca has been reported to have a good safety record with no serious side-effects. Countries might think it is a safer choice with a lower risk. However, problems associated with Astrazeneca have been surfacing, causing some countries to abandon it.
Correlation between effectiveness and acceptance
Although the effectiveness of Astrazeneca has not changed since it was approved by the government, the acceptance rate of the vaccine has deteriorated. This further proves that the effectiveness of the vaccine is not the only factor that is taken into account when deciding between vaccines (16). From the graph below, it is clear to see that the acceptance of the Pfizer vaccine remains significantly higher than the acceptance rate of Astrazeneca. It is clear to see that there is a slightly positive correlation between the effectiveness of the vaccine and its acceptance rate as Pfizer’s efficacy rate is higher than that of Astrazeneca, and as shown in the graph below, Pfizer is more widely accepted internationally (15).
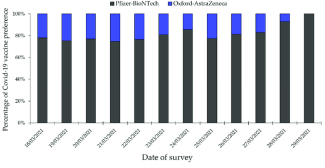
Figure 3: Different COVID-19 vaccines’ distribution around the world
When an individual is choosing the type of vaccine they want administered, they consider many factors other than the effectiveness, such as the availability of the vaccines. Some vaccines could be more available in some countries than others. For example, Pfizer is used more internationally, while the distribution of Astrazeneca is largely more focused on Europe. This means that people living on other continents would be less aware of this vaccine and so more likely to choose to have the Pfizer vaccine. Some people are slightly concerned about the side effects, and this leads them to prefer one vaccine over the other.
The rollout of Astrazeneca was temporarily stopped for a short period as it was found to be causing blood clots in some individuals. This led some people to prefer Pfizer over Astrazeneca, as it seemed to have less serious side-effects. Likewise, there are regulations and incentives that encourage residents to choose a particular vaccine, like lottery systems, vaccine mandates and restrictions for people taking unauthorised vaccines. The data collected for the number of vaccinations might not be accurate as some might take the vaccine just to receive the benefits but not because the acceptance rate has increased (15).
Discussion
Since 2020, when the COVID-19 pandemic began, many vaccines have been created in an attempt to successfully achieve herd immunity. The fact that there are so many vaccines means that the general public is able to choose which one they want administered, and how they choose one vaccine over the others depends on many factors.
From this research, we have found that the effectiveness of the vaccines does affect the acceptance rate, as some people prefer to have the more effective vaccine, concluding it is more likely to keep them safe from this virus. Despite there being a positive correlation, however, it is not overwhelmingly significant, which suggests that there are other factors that affect the acceptance rate.
Other major factors that affect the acceptance rate are the area in which the individuals live and also their age. Different countries are rolling out different vaccines. This could potentially be an unsuitable way in which vaccines are administered as the most effective vaccines, which could save the most lives, would be neglected. Thus one of the important conclusions to be drawn here is that knowledge of vaccines should be more widely available; having more knowledge would allow each individual to make their own informed decision about which vaccine they deem suitable. However the caveat to this is that perhaps some vaccines are less available in some areas and some have storage difficulties, such as Pfizer which needs to be stored in extremely cold conditions.
Different age ranges may also have their own preference towards the vaccine, which may be less informed, causing subconscious bias and leading to people choosing a less effective vaccine. An example of this is the trust that an older generation may have towards live attenuated vaccines. This trust, which they have built from past experience such as the Spanish Flu pandemic, may incline them to choose the Astrazeneca vaccine. This means that they don’t choose Pfizer, which has a higher efficacy, because of the way it works, which they do not recognise as a plausibly effective technique due to its modernity.
Another major factor which affects acceptance rate is the misinformation which is common in the media. As people understand the side effects of the vaccine, especially the much rarer ones, some people reason that the supposed, highly exaggerated danger of taking a vaccine outweighs the protection it offers. For example, the rare instance of vaccine-induced thrombosis with thrombocytopenia syndrome has scared people into worrying about the danger of the Astrazeneca vaccine. Perhaps it is this misinformation that is most detrimental to the global vaccine rollout.
Bibliography:
- “COVID Live – Coronavirus Statistics.” Worldometer, 2020, https://www.worldometers.info/coronavirus/. Accessed 16 April 2022.
- “Autoimmune phenomena following SARS-CoV-2 vaccination – PMC.” NCBI, 10 July 2021, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8270741/. Accessed 30 April 2022.
- “Understanding How Vaccines Work.” CDC, August 2018, https://www.cdc.gov/vaccines/hcp/conversations/understanding-vacc-work.html. Accessed 17 April 2022.
- Bono, Suzanna Awang, et al. “Factors Affecting COVID-19 Vaccine Acceptance: An International Survey among Low- and Middle-Income Countries.” vol. Vaccines, vol. 9, no. 5, 1 May 2021, p. 515, www.mdpi.com/2076-393X/9/5/515, 10.3390/vaccines9050515. Accessed 4 July 2021.
- UK government. “Regulatory approval of Pfizer/BioNTech vaccine for COVID-19.” GOV.UK, 2 December 2020, https://www.gov.uk/government/publications/regulatory-approval-of-pfizer-biontech-vaccine for-covid-19. Accessed 13 April 2022.
- “Coronavirus (COVID-19) Vaccinations.” OurWorldInData.org, 2020, https://ourworldindata.org/covid-vaccinations. Accessed 22 April 2022.
- “What are mRNA vaccines and how do they work?” MedlinePlus, 23 March 2022, https://medlineplus.gov/genetics/understanding/therapy/mrnavaccines/. Accessed 23 April 2022.
- “Comparing the longer-term effectiveness of a single dose of the Pfizer-BioNTech and Oxford-AstraZeneca COVID-19 vaccines across the age spectrum.” The Lancet, 12 March 2022, https://www.thelancet.com/journals/eclinm/article/PIIS2589-5370%2822%2900074-8/fulltext.
- “Effectiveness of the Pfizer-BioNTech and Oxford-AstraZeneca vaccines on covid-19 related symptoms, hospital admissions, and mortality in older adults in England: test negative case-control study.” BMJ, 13 May 2021, https://www.bmj.com/content/373/bmj.n1088.full.
- “Vaccine side-effects and SARS-CoV-2 infection after vaccination in users of the COVID Symptom Study app in the UK: a prospective observational study.” NCBI, 21 July 2021, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8078878/. Accessed 30 April 2022.
- WHO (2020). Coronavirus disease (COVID-19): Herd immunity, lockdowns and COVID-19. [online] www.who.int. Available at: https://www.who.int/news-room/questions-and-answers/item/herd-immunity-lockdowns-and-covid-19.
- “Herd Immunity: What Is It and Can It End The Coronavirus Pandemic?” WebMD, 3 February 2022, https://www.webmd.com/lung/what-is-herd-immunity#2-5. Accessed 2 May 2022.
- www.astrazeneca.com. (n.d.). AZD1222 US Phase III primary analysis confirms safety and efficacy. [online] Available at: https://www.astrazeneca.com/media-centre/press-releases/2021/azd1222-us-phase-iii-primary-analysis-confirms-safety-and-efficacy.html.
- Pfizer (2020). Pfizer and BioNTech Conclude Phase 3 Study of COVID-19 Vaccine Candidate, Meeting All Primary Efficacy Endpoints | Pfizer. [online] Pfizer.com. Available at: https://www.pfizer.com/news/press-release/press-release-detail/pfizer-and-biontech-conclude-phase-3-study-covid-19-vaccine.
- Sallam, Malik. “COVID-19 Vaccine Hesitancy Worldwide: A Concise Systematic Review of Vaccine Acceptance Rates.” no. Vaccines, vol. 9, no. 2, 16 Feb. 2021, p. 160, www.mdpi.com/2076-393X/9/2/160/htm, 10.3390/vaccines9020160.
- Kaplan, Robert M., and Arnold Milstein. “Influence of a COVID-19 Vaccine’s Effectiveness and Safety Profile on Vaccination Acceptance.” no. Proceedings of the National Academy of Sciences, vol. 118, no. 10, 9 Mar. 2021, www.pnas.org/content/118/10/e2021726118, 10.1073/pnas.2021726118.
- KATELLA, KATHY. “Comparing the COVID-19 Vaccines: How Are They Different?” Yale Medicine, 30 March 2022, https://www.yalemedicine.org/news/covid-19-vaccine-comparison. Accessed 2 May 2022.
- “COVID-19 Vaccine AstraZeneca confirms 100% protection against severe disease, hospitalisation and death in the primary analysis of Phase III trials.” AstraZeneca, 3 February 2021, https://www.astrazeneca.com/media-centre/press-releases/2021/covid-19-vaccine-astrazeneca -confirms-protection-against-severe-disease-hospitalisation-and-death-in-the-primary-analysis -of-phase-iii-trials.html. Accessed 2 May 2022.




